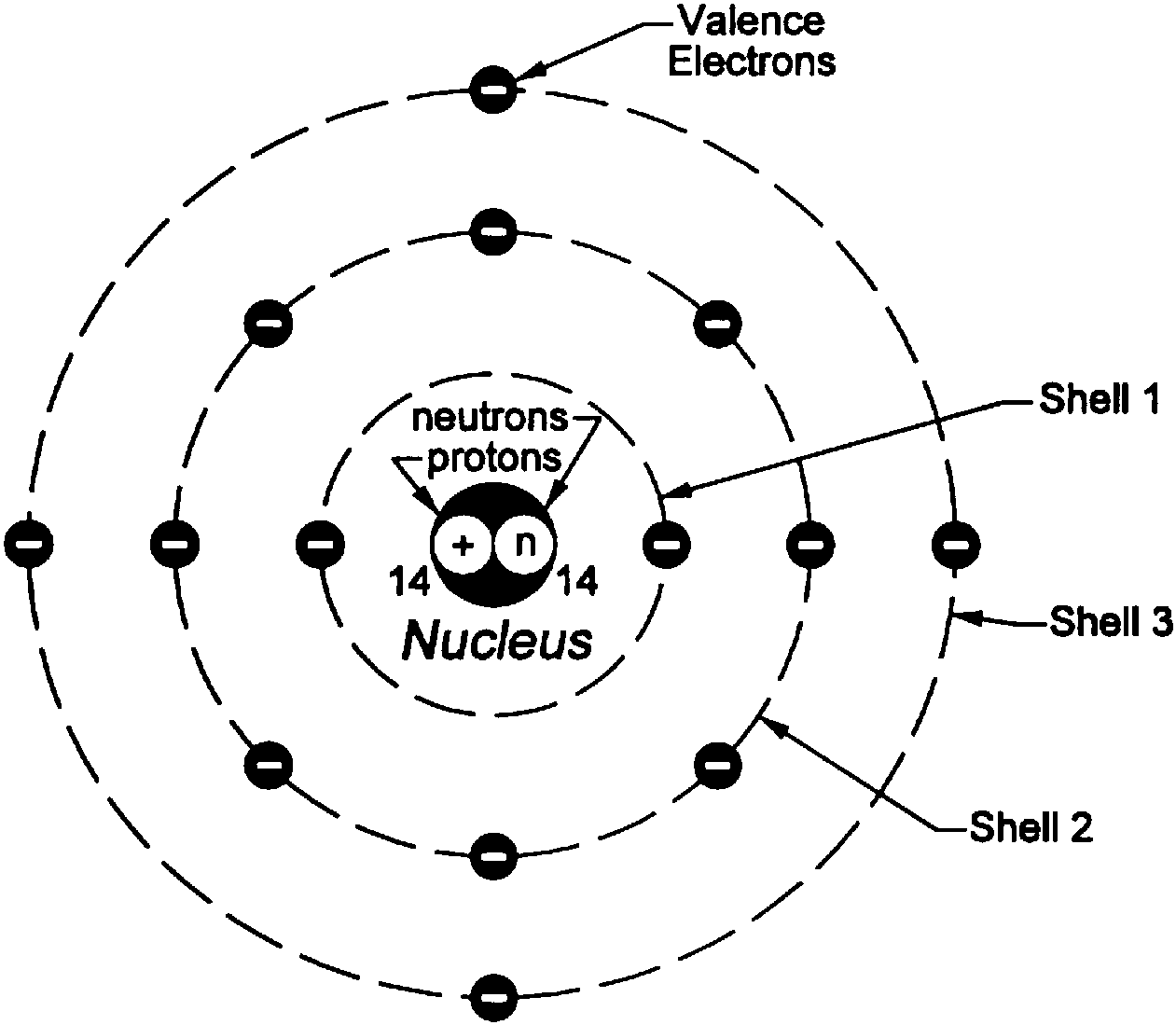
Let him throw stones at the wall from a distance. Let a child stand in front of a wall with his eyes closed. Let us think of an activity in an open field to understand the implications of this experiment. In the words of Rutherford, “This result was almost as incredible as if you fire a 15-inch shell at a piece of tissue paper and it comes back and hits you”. Surprisingly one out of every 12000 particles appeared to rebound.Some of the α-particles were deflected by the foil by small angles.Most of the fast moving α-particles passed straight through the gold foil.Since the α-particles were much heavier than the protons, he did not expect to see large deflections.īut, the α-particle scattering experiment gave totally unexpected results (Fig.2). It was expected that α-particles would be deflected by the sub-atomic particles in the gold atoms.Since they have a mass of 4 u, the fast-moving α-particles have a considerable amount of energy. α-particles are doubly-charged helium ions.This gold foil was about 1000 atoms thick. He selected a gold foil because he wanted as thin a layer as possible.In this experiment, fast moving alpha (α)-particles were made to fall on a thin gold foil. Rutherford designed an experiment for this. So, the atom as a whole is electrically neutral.Īlthough Thomson’s model explained that atoms are electrically neutral, the results of experiments carried out by other scientists could not be explained by this model, as we will see below.Įrnest Rutherford was interested in knowing how the electrons are arranged within an atom. The negative and positive charges are equal in magnitude.An atom consists of a positively charged sphere and the electrons are embedded in it.We can also think of a watermelon, the positive charge in the atom is spread all over like the red edible part of the watermelon, while the electrons are studded in the positively charged sphere, like the seeds in the watermelon (Fig. The electrons, in a sphere of positive charge, were like currants (dry fruits) in a spherical Christmas pudding. Thomson proposed the model of an atom to be similar to that of a Christmas pudding. Thomson was the first one to propose a model for the structure of an atom. Let us discuss those various models to explain how these particles are arranged within the atom. For explaining this, many scientists proposed various atomic models. It was then considered necessary to know how electrons and protons are arranged within an atom. But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of Dalton’s atomic theory. We have learnt Dalton’s atomic theory in previous lesson, which suggested that the atom was indivisible and indestructible.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
